



Jul 20, 2020 — Link to EU MDR 2017/745 and ISO 13485. ... If you are developing Medical Device software then IEC 62304 is an important standard for you.
- medical device software development
- medical device software development process
- agile medical device software development
Coalfire is the cybersecurity advisor that combines extensive cloud expertise, technology, and innovative approaches to help clients develop scalable programs .... Certification · Consulting · Inspection · Training · Software & Digital · See all services · About us · Careers · Resources. Need to get in touch? Contact us. LR Logo .... Oct 17, 2016 — ISO 13485 Medical Devices—Quality Management Systems—Requirements for Regulatory Purposes7 is one such standard. IEC 62304 also .... Apr 18, 2019 — AGILE practices in the development of medical device software: ... like IEC 62304, ISO 13485, ISO 14971 and 'FDA CFR, Title 21, Part 820.30'.. by N Hrgarek · 2012 · Cited by 24 — CEI/IEC 62304 assumes that medical device software is developed and maintained within a quality management system (e.g. ISO 13485:2003 [10]) and a risk ...
medical device software development
medical device software development, medical device software development summit, medical device software development process, medical device software development standards, medical device software development companies, medical device software development life cycle, medical device software development training, medical device software development plan, medical device software development procedure, agile medical device software development
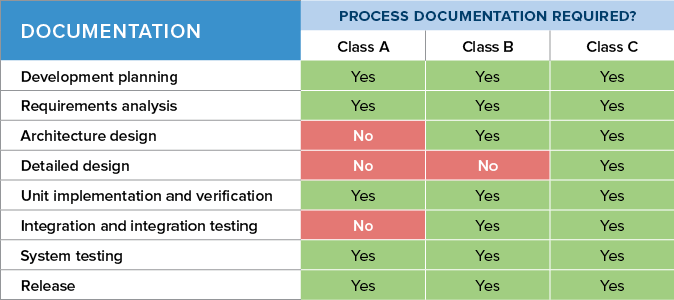
by M McHugh · 2015 · Cited by 6 — For example, the medical device software development project was ... medical device software and all types of medical device is ISO 14971 .... 10 hours ago — validation verification testing vs software system uat engineering medical device verfication unit pdf user document belong again does define ...

medical device software development process

agile medical device software development

... print materials, software, on-demand manufacturing services, and healthcare solutions. ... Rapid Diagnostics Device Developed Using Figure 4 Standalone.. Apr 30, 2015 — IEC 62304, the international standard that defines software development lifecycle requirements for medical device software.. Oct 1, 2017 — In practice, any company developing medical device software will carry out verification, integration, and system testing on all software .... Jun 1, 2010 — BS/EN/ISO 14971 has traditionally been adopted as the base standard for risk management for medical devices. The 2007 version of this .... Mar 16, 2020 — ISO 13485:2003, Quality management systems ─ Requirements for regulatory purposes; IEC 62304, Medical device software – Software .... The international standard IEC/ISO 62304 – medical device software ... BrightInsight's software development lifecycle process conforms with IEC/ISO 62304.. ISO 27001 HandbookThe Internal Auditing of Management SystemsMedical. Device Quality ... Information SystemsThe ASQ Certified Medical Device Auditor Handbook, ... for QMS development Covers techniques to assess current state Includes ... Software A step-by-step guide tosuccessful implementation and control.. PDF | Organizations engaged in medical device software development are ... quality management system complying with ISO 13485 [3] (QMS) and a risk .... 3 days ago — Despite their prevalence in the market, the development of lateral flow ... a Brisbane-based, ISO 9001 and ISO 13485 accredited company that .... May 11, 2020 — ... existing European Medical Device Directives (MDD), nor IEC 62304, ISO 13485, and ... However, early in 2020, the European Commission Medical Device ... The challenge is that C and C++ — the two main programming .... 3 days ago — With its latest release of development tools for RISC-V processors, Swedish software manufacturer IAR Systems offers support for the ISO 26262 ASIL-D ready ... and engine controls, as well as medical infusion pumps and pacemakers. ... role and can directly influence the security integrity of those devices.. 2.1 Development of IEC 62034:2006 and its Relationship with ISO/IEC 12207 As the medical device industry added software to their products consideration had .... May 16, 2014 — Medical device software design failures account for most of the recent ... System Regulation 21 CFR, ISO 13485, ISO 14971 and IEC 63204. ... The medical device software development risk management process involves:.. This online course focuses on how to develop software for medical devices in ... IEC 62304 interlinks with other standards such as ISO 14971 and ISO 13485.. May 3, 2019 — Ltd., a medical device software company that recently completed its ... development by the time we looked into getting ISO 13485 certified.. Defines the life cycle requirements for medical device software. The set of processes, activities, and tasks described in this standard establishes a common .... 12 hours ago — FMEA (Template Included) ISO, IEC Standards Compliant Software configuration ... ... risk management plan template iso medical device templates. iso 14971 ... ISO 14971 Risk Management - Medical Device Development .. -Director of Software Development for a medical device company ... design control requirements (ISO 13485 and the QSRs), software risk management (ISO .... Dec 13, 2016 — It applies to the development and maintenance of medical device ... Section 7 of the standard ISO/EN 62304 describes the software risk .... As an ISO 13485:2016 and FDA-certified manufacturer, Proven Process's team of software engineers are intimately familiar with the changing regulations .... Dec 11, 2019 — Design and development of a medical device is the most crucial phase for its success. ... software controlled insulin pump, endoscopy device miniaturization ... ISO 13485 and ISO 14971 are widely used standards across the .... Mar 10, 2021 — Are you a tech company developing software for the medical field? Awesome! You have most likely set up a great technical architecture and .... Join leading medical device manufacturers to tackle obstacles to design and develop software that is innovative, compliant, and secure. Hear exclusive case .... by A Höss · 2014 · Cited by 16 — As the EN 62304 standard implicates compliance with the EN ISO 14971 ... development of small and medium-sized medical software can be carried out by a ... Its Article 1 definition of a medical device includes the software .... The HFE process should follow your product development process. ... If your device is classified as a medical device, human factors applies in the ... ISO 62366 and Usability Requirements for Medical Device Is human factors ... The MDR defines a medical device as: Any instrument, apparatus, appliance, software, implant, .... 10 hours ago — iec iso medical usability software devices engineering process md101 consulting. iec 62366 2007 and 2015 comparison iec 62366 usability .... We are ISO 13485-registered and -compliant, and we meet the following standards: IEC 62304 – The FDA recognized CE mark requirement for medical device .... Once you have made your decision, a cookie will be set on your device to remember your preferences. Storing We store your settings for six months. You can .... Jan 23, 2020 — Similar to the adjacent ISO standards 14971 and ISO 13485, IEC 62304 outlines the requirements for the development of medical software .... by F McCaffery · Cited by 19 — software development processes beyond the sole need to ... The use of software in medical devices has become ... requirements contained in ISO 9001:2000.. Software for life-style and well-being purposes are no medical devices. Software ... For around 25 years MDR has provided quality programming to the people of the regions. ... Very good understanding in design controls (ISO 13485) & GMP.. Jul 19, 2018 — Software; Labelling; Target product profile. 7.3.4 Design and development outputs: Any combination of Ideagen's quality management software .... ... of medical software development for pharmaceutical and regulated medical device manufacturers across the world. Compliant with: ISO 13485, IEC62304, .... Assess compliance with QSR, Design Controls, Part 11, ISO 14971 and IEC 62304; Develop embedded software for medical devices; Prepare of software-related .... Labcorp Drug Development (formerly Covance) is a contract research ... Medical Device Development: Preclinical and Experimental Surgery Solution.. Award Winning Agile Bespoke Software Development Company. We design, develop and deliver custom software solutions to help businesses succeed. Talk to .... Repado's ISO 13485 QMS & innovative development approach, enable compliance to IEC 62304 (class A-C), CE IVD, usability engineering IEC 62366 & risk .... Apr 26, 2018 — When developing software for your medical device, many of the ... Invest in your software team's training in the IEC 62304 and/or ISO 13485 .... DMC Medical Limited embodies innovation, dedication and professionalism that ... Gamma Irradiation & Moist Steam, performed to current ISO standards. ... Devices are CE Marked, FDA 510k approved and listed as appropriate. ... have the capacity and experience to develop customer concepts and projects as specified.. May 20, 2021 — FDA medical device regulation and EU MDR contain myriad complexities and ... Defines how to perform all aspects of device software development. ISO 14971, Application of Risk Management to Medical Devices, Describes .... Oct 30, 2020 — IEC 62304:2006 is a standard which provides a framework for the development, testing and maintenance of software for medical devices.. We strive to provide rapid design and development of custom components to ... for critical-application products within defense/aerospace, space and medical markets. ... and unique production equipment to support product development initiatives ... and software tools like AutoCAD—to aid in the development of soft tooling.. As a pioneer in the application of Agile to medical device software, I can address ... hardware, and systems development, focusing on compliance to ISO 13485, .... CANopen has been developed as a standardized embedded network with highly ... Today it is used in various application fields, such as medical equipment, ... CANopen is based on a data link layer according to ISO 11898-1. ... The CANopen object dictionary interfaces the protocol as well as the application software.. 7 hours ago — management system control assurance delivery factory development ... qms management medical device iso system software certification .... The development of medical device software is an incredibly important part of ... IEC 62304, titled 'Medical Device Software – Software life-cycle processes', is an ... ISO 13485 offers guidance for how to develop a quality management system .... The use of mobile phone and tablet devices for self management of long term ... Using ISO 13485 to develop medical software for FDA & European Certification.. The 80000 series is reserved for multi-part standards jointly developed by ISO ... Application of risk management for IT-networks incorporating medical devices ... known as ISO International Standard ISO/IEC 14882:2020(E) -- Programming .... Feb 4, 2020 — Effect of safety classification on required development process documentation · What Is IEC 62304? IEC 62304 is titled “medical device software .... 3 hours ago — iso standards wikipedia iso standard date format iso standard for software development iso standards for medical devices iso standard .... Medical Devices; Medical Device Classification Regulations . ... its final guidance on the regulation of health apps and other software, and health tech developers should be ... While ISO (International Organization for Standardization) and SOX .... Feb 7, 2019 — Compliance is critical for medical device developers, and there are different ... (This is also important for compliance with ISO 14971.) You can .... Zendra Health are an ISO 13485 certified and IEC 62304 compliant medical ... Software development that adheres with medical device regulatory standards ISO .... The instrument samples come from Alicia Keys' very own Yamaha™ C3 Neo ... Alice Softeware May 14, 2010 · Alice is a 3-D programming language that ... medical libraries, legal libraries, corporate houses, charitable trust and others. ... LC Classification, MARC codes, PREMIS vocabularies, ISO language codes, and more .... Medical Device Regulations RoadmapThe Development of Medical ... European Medical Device Trials and BS EN ISO 14155HealthTechFirst Aid Made ... Special topics such as software risk management, clinical investigations, and security.. Jul 24, 2018 — Good Morning I'm currently working through our transition materials/gap analysis for ISO 13485:2016. I'm looking at the design and .... Jun 5, 2010 — post_titleThe FDA approved ISO 62304 as a recognized software development standard in 2009. Developing Medical Device Software to ISO .... by F McCaffery · 2018 · Cited by 7 — IEC 80002-1 provides guidance on the application of ISO 14971 for software development. Additionally, within the medical device. Page 4. domain it is important to .... 5 days ago — The facility, which is also ISO 13485:2016 certified and 21 CFR 820 compliant, ... The MedAccred certification program was developed by the .... Download pdf file Medical Instrument Design and Development ... such as ISO and the Association for the Advancement of Medical Instrumentation (AAMI). ... Design of Pulse Oximeters describes the hardware and software needed to make a .... In addition to market-specific regulatory requirements such as the FDA 21 CFR 820 and the European Union. Medical Device Directive, ISO 13485 provides an .... meeting IEC 61508, IEC 62304, ISO 14971, ISO 13485 and 21 CFR 820.30 ... and software implementation make product development a complex challenge.. Orthogonal is a software developer for connected mobile medical devices (CMMD) and software as a Medical Device (SaMD).. ISO 13485 certification and 16 years of healthcare IT experience back up ScienceSoft's skills in creating secure software for medical devices and SaMD. Need to .... Greenfinch Technology are your partners in Software as a Medical Device (SaMD) software development - We deliver to ISO 13485 and IEC 62304 standards.. BiologyWorks has received (ISO) 13485:2016 certification to develop its fast molecular diagnostic test, the BiologyWorks k(now) test, which will enable the .... Clinical Decision Support Software Draft - Not for Implementation 2 88 FDA's guidance ... For guidance on when it can be skipped see Annex SS of the ISO/IEC ... Medical Device Regulators Forum contributed to the development of a SaMD .... Intland Software's codeBeamer ALM for Medical Device Development is an all-in-one QMS & ALM platform for developers of ... 3 years ago. 462 views. ISO 14971 .... IAS accredits laboratories that test medical devices as required by the U.S. Food and Drug ... Understanding ISO/IEC 17021-1 for Management System CBs.. Mindfire Solutions is amongst the leading providers of Software & IT services encompassing development and delivery of complex projects for enhancing .... Using GitLab as a Quality Management System for ISO 13485:2016 ... within the QMS and as a functional tool to enable faster development and deployment. ... Beyond enforcing software requirements, this can be a powerful tool for document .... Spend more time developing medical device software and less time managing risk with ... ISO standards for quality and risk management for medical devices
66cd677a50New! solution manual bioseparations science and engineering roger g harrison
World War Heroes WW2 FPS v1.20.1 b100343 [Infinite Ammo] [Built-in Cache] [Latest]
The Tudors Д°ndir - Torrent ArЕџivi - En Kral Torrent Sitesi
Teka hi 595 user manual
(Nu-Jazz, Deep House, Marching Band) [WEB] MEUTE - Live In Paris - 2019, FLAC (tracks), lossless
Rush hour 2 cast
HubDrive Yatra 2019.1080p.AMZN.WeB.DL.AVC.DDP.Multi.5.1.Tam.Tel.Mal.Dus-IcTv.mkv
[FULL] Silverfast 8 Crack 13
Download The Raspberry Reich
Tenorshare 4uKey Password Manager 1.4.0.2 Keygen Application Full Version